Hypertrophic Obstructive Cardiomyopathy (HOCM)
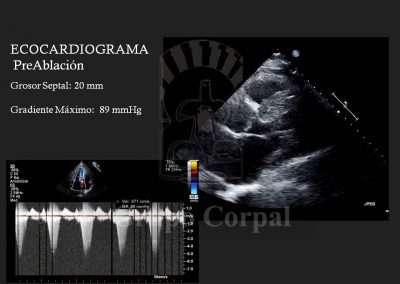
Hypertrophic Obstructive Cardiomyopathy (HOCM) is a serious cardiomyopathy that is very difficult to treat. It can be progressive, present without a genetic component or be transmitted genetically. The physiopathological base consists of asymmetric hypertrophy with disarrangement of fibres, primarily in the interventricular septum, that dynamically obstructs the outflow of the left ventricle; when ventricular contraction progresses during systole, the hypertrophic septum moves towards the outflow and pulls the anterior mitral leaflet to the ventricular outlet. This can also cause mitral closure problems, leading to mitral valve insufficiency.
We know that the severity of the obstruction is associated to serious cardiac events such as malignant arrhythmia, sudden death, heart failure and acute pulmonary oedema. Medical treatment consists of the administration of drugs that reduce the heart’s contractile power, such as beta blockers and calcium antagonists, but they rarely lead to significant reductions in the sub-aortic gradient. Surgery has made important contributions in an attempt to reduce the obstruction, resecting part of the hypertrophic septum (myectomy) or making cuts in the septum to prevent contraction of the treated area (myotomy). These are, however, palliative measures. Some authors have also recommended mitral valve replacement, and others suggest the implantation of a pace maker that changes the origin of ventricular contraction, which can reduce the pressure gradient.
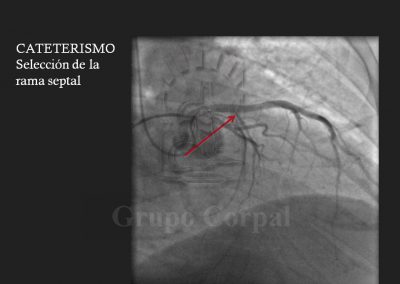
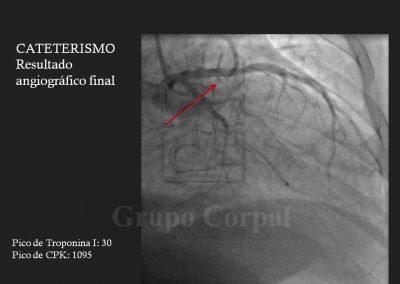
A highly original and effective percutaneous method for the palliative treatment of this condition has been developed in the last 20 years. It consists of creating a myocardial infarction located in the inter-ventricular septal area that generates a dynamic obstruction. The infarcted myocardium does not contract, so it reduces the degree of obstruction, and even the mitral insufficiency can be solved. The key to the treatment lies in limiting the controlled infarction to the hypertrophic septal area that causes the problem, so great precision is required when selecting the target. This requires familiarity with the vascularisation of the interventricular septum. There is usually a bunch of septal vessels that irrigate the septum, although the anatomy varies. A CT-64 study shows us the septal coronary anatomy, which helps us to design the treatment. From a technical perspective, 2 arterial and 1 venous access are required. One of the former is for monitoring pressure in the left ventricle during the procedure, and for pre- and post-treatment ventriculographies. The venous line is to measure pulmonary pressure and for the implantation of an electrode catheter in the right ventricle as a prophylactic pace maker, as transient A-V block can present during septal ablation.
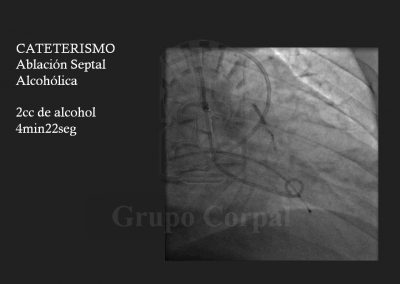
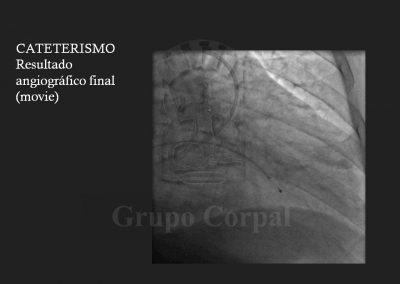
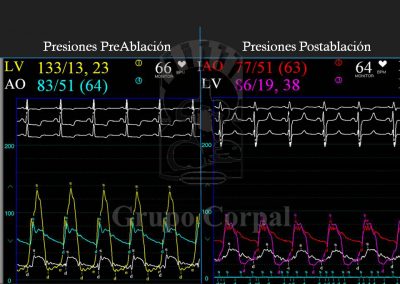
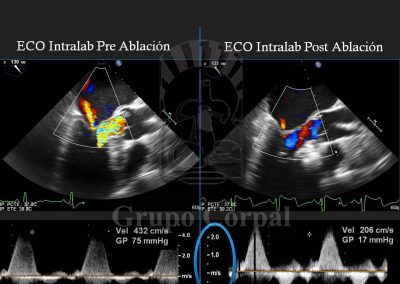
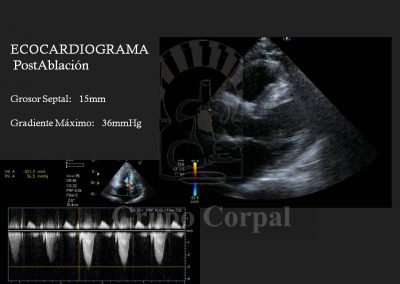
Finally, the second arterial line is for a guide catheter with lateral orifices that measured both systemic pressure and left ventricle pressure at all times, thus monitoring the sub—valvular gradient throughout the procedure. The guide catheter in the left coronary ostium enables the insertion of a long guide in the septal bunch, branch of the anterior descending artery, passing a balloon-catheter of the same diameter to generate complete occlusion; when verified, the balloon remains inflated and the guide is removed. The free access is used to slowly diffuse 100 degree alcohol to cause abrasion of the irrigated area and, therefore, localised infarction of the target zone. This is immediately followed by the reduction or disappearance of the sub-valvular gradient and the degree of mitral insufficiency. These aspects are haemodynamically and angiographically evaluated at the end of the procedure. There is usually immediate clinical benefit and prompt discharge.